Introduction: Venetoclax (VEN), a novel BCL-2 inhibitor, received accelerated FDA approval for the treatment of newly diagnosed (ND) acute myeloid leukemia (AML) in adults aged 75 years or older, or who have comorbidities precluding use of intensive chemotherapy, in combination with hypomethylating agents (HMAs: azacitidine [AZA] or decitabine [DEC]), or low-dose cytarabine (LDAC). As Phase III trials have demonstrated relative efficacy of VEN combinations, this real-world initiative provides insight into the characteristics and outcomes of ND AML patients (pts) treated with VEN combinations vs non-VEN-based regimens in clinical practice.
Methods: The AML Real world evidenCe (ARC) Initiative is a multicenter chart review study of adult pts with ND AML treated with VEN ≥ 4-11-2016 (VEN cohort) or non-VEN-based regimens ≥ 5-15-2015 (control cohort) from different academic sites in the US. Pts in the control cohort were matched to pts in the VEN cohort based on age (<60; 60-74; ≥75) and ELN risk. Interim descriptive results are presented from data as of 6-26-2020; data collection is ongoing, with a targeted sample size of over 500 pts. First and best responses to therapy, overall survival (OS), and event-free survival (EFS) were analyzed. A subgroup of pts receiving high-intensity chemotherapy regimens in the control cohort and their matched pts in the VEN cohort was analyzed separately (high-intensity therapy matched subgroup).
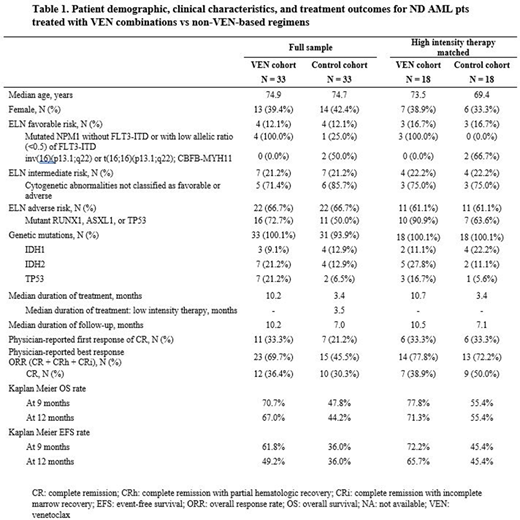
Results: At time of interim data analysis, 33 VEN and 33 control ND AML pts were included. Combinations in the VEN cohort included HMA (AZA [26 pts, 78.8%], DEC [6 pts, 18.2%]) or LDAC (1 pt, 3.0%); the control cohort received high- (18 pts, 54.5%) or low-intensity regimens (15 pts, 45.5%; HMA: 12 pts; enasidenib: 2 pts; LDAC: 1 pt). Median age was 75 years in both VEN and control cohorts, 39.4% and 42.4% were female, respectively, and 63.6% had de novo AML in both cohorts. The majority had ECOG performance status of 1 (VEN: 66.7%; control: 63.3%) and adverse-risk ELN classification (66.7% in both cohorts). All pts with favorable-risk in the VEN cohort (100.0%) had mutated NPM1 without FLT3-ITD or with low allelic ratio (<0.5) of FLT3-ITD, while 50.0% of pts with favorable-risk in the control cohort had inv(16)(p13.1;q22) or t(16;16)(p13.1;q22); CBFB-MYH11 (Table 1). IDH1 and IDH2 mutations were present in 9.1% and 21.2%, respectively, in the VEN cohort and 12.9% and 12.9% in the control cohort; TP53 mutation was present in 21.2% and 6.5% of the VEN and control cohort, respectively.
In the high-intensity therapy matched subgroup (VEN: n=18; control: n=18), median age was 74 and 69 in VEN versus control, respectively. The majority of pts had ECOG performance status of 1 (VEN: 76.5%; control: 75.0%) and adverse-risk ELN classification (61.1% in both cohorts). High-intensity treatment regimens used for the control group were: CPX-351 (n=10; 55.6%), 7+3 (n=4; 22.2%), 7+3 + gemtuzumab ozogamicin (n=3; 16.7%), cytarabine + mitoxantrone (n=1; 5.6%). In the VEN matched high-intensity subgroup, 15 (83.3%) received VEN + AZA, 2 (11.1%) received VEN + DEC, and 1 (5.6%) received VEN + LDAC. Ten (55.6%) initiated VEN as an inpatient treatment.
Outcomes for the full sample and subgroup are presented in Table 1. In the full sample, the overall response rate (ORR: CR+CRi+CRh) was 69.7% in the VEN cohort and 45.5% in the control cohort. The 1-year OS was 67.0% in the VEN cohort and 44.2% in the control cohort for the full sample; and 71.3% and 55.4%, respectively, in the high-intensity therapy matched subgroups.
Conclusions: The ongoing ARC Initiative demonstrates real-world treatment effectiveness of VEN combinations in clinical practice. The real-world outcomes for VEN combinations for ND AML pts appear consistent with results reported from clinical trials, though a small number of pts were assessed. Further analyses of comparisons to matched controls, both overall and in the high-intensity therapy matched subgroup, and continuing data collection will allow a more robust assessment of the comparative effectiveness of VEN combinations in the real-world setting.
Pollyea:Syndax: Consultancy; Syros: Consultancy; Novartis: Consultancy; Karyopharm: Consultancy; Janssen: Consultancy; 47: Consultancy, Research Funding; Genentech: Consultancy; Takeda: Consultancy; Celgene/BMS: Consultancy; Agios: Consultancy; Glycomimetics: Other; Amgen: Consultancy; Abbvie: Consultancy, Research Funding; Daiichi Sankyo: Consultancy; Pfizer: Consultancy. Talati:BMS: Honoraria; Jazz: Speakers Bureau; AbbVie: Honoraria; Pfizer: Honoraria; Astellas: Speakers Bureau. Lai:Abbvie: Consultancy; Jazz: Speakers Bureau; Astellas: Speakers Bureau; Macrogenics: Consultancy; Agios: Consultancy. Abedin:Helsinn Healthcare: Honoraria; Agios: Honoraria; Jazz Pharmaceuticals: Honoraria; Pfizer: Research Funding; Helsinn Healthcare: Research Funding; Actinium Pharmaceuticals: Research Funding. Zeidner:Sumitomo Dainippon Pharma Oncology, Inc.: Research Funding; Merck: Research Funding; Forty-Seven: Other: Travel Reimbursement, Research Funding; AROG: Research Funding; AsystBio Laboratories: Consultancy; Daiichi Sankyo: Honoraria; Takeda: Consultancy, Honoraria, Other: Travel Reimbursement, Research Funding; Pfizer: Honoraria; Genentech: Honoraria; Celgene: Consultancy, Honoraria, Research Funding; AbbVie: Honoraria, Other: Independent Review Committee; Agios: Honoraria. Xavier:Kite/Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; AstraZeneca: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Morphosys: Honoraria, Membership on an entity's Board of Directors or advisory committees; TG therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees; Verastem: Honoraria, Membership on an entity's Board of Directors or advisory committees; Acrotec: Consultancy, Speakers Bureau; Biegene: Speakers Bureau; Genentech: Speakers Bureau; Seattle Genetics: Speakers Bureau; Celgene/BMS: Speakers Bureau; Epizyme: Speakers Bureau; Astellas: Speakers Bureau; Incyte: Speakers Bureau. Wolach:AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Fees for lectures and Consultancy, Research Funding; Pfizer: Consultancy, Honoraria; Janssen: Other: Fees for lectures and Consultancy; Amgen: Other: Fees for lectures and Consultancy; Astellas: Consultancy, Honoraria, Other: Fees for lectures and Consultancy; Novartis: Consultancy, Honoraria, Other: Fees for lectures and Consultancy. Moshe:Novartis: Membership on an entity's Board of Directors or advisory committees, Other: Lecturer; Astellas: Membership on an entity's Board of Directors or advisory committees, Other: lecturer; AbbVie: Membership on an entity's Board of Directors or advisory committees, Other: Fee for lec. Choi:AbbVie: Current Employment, Current equity holder in publicly-traded company. Bui:AbbVie: Current Employment, Current equity holder in publicly-traded company. Svensson:AbbVie: Current Employment, Current equity holder in publicly-traded company. Kye:AbbVie: Current Employment, Other: may hold stock or other options. Burne:Analysis Group, Inc., which has received consultancy fees from AbbVie: Current Employment. Guerin:Abbvie: Consultancy, Other; Sanofi Genzyme: Consultancy, Other: Annie Guerin is an employee of Analysis Group, Inc. which received consultancy fees from Sanofi Genzyme.; Novartis Pharmaceuticals Corporation: Consultancy, Other: Annie Guerin is an employee of Analysis Group, Inc. which received consultancy fees from Novartis.. Ma:Genentech, Inc.: Current Employment, Current equity holder in publicly-traded company. Montez:Genentech, Inc.: Current Employment; F. Hoffmann-La Roche: Current equity holder in publicly-traded company. Goldberg:Dava Oncology: Honoraria; Pfizer: Research Funding; Celularity: Research Funding; AROG: Research Funding; Aprea: Research Funding; ADC Therapeutics: Research Funding; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Consultancy; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Aptose: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal